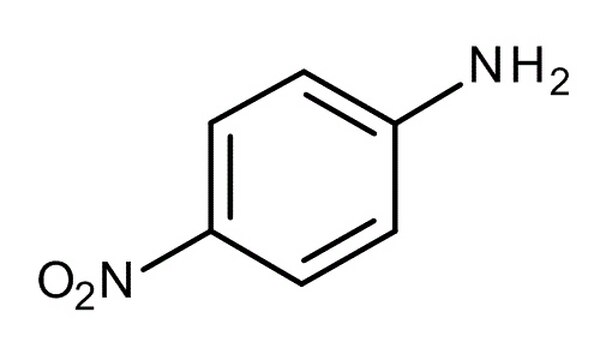
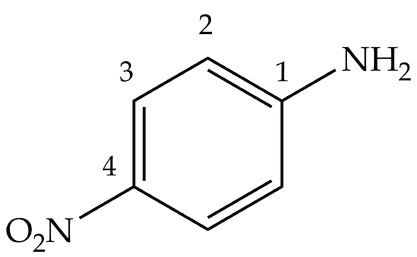
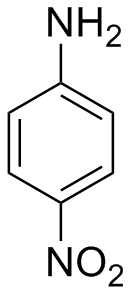
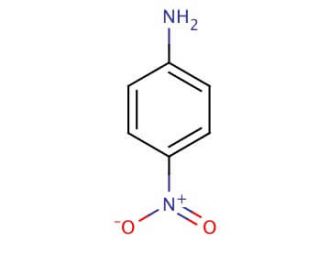
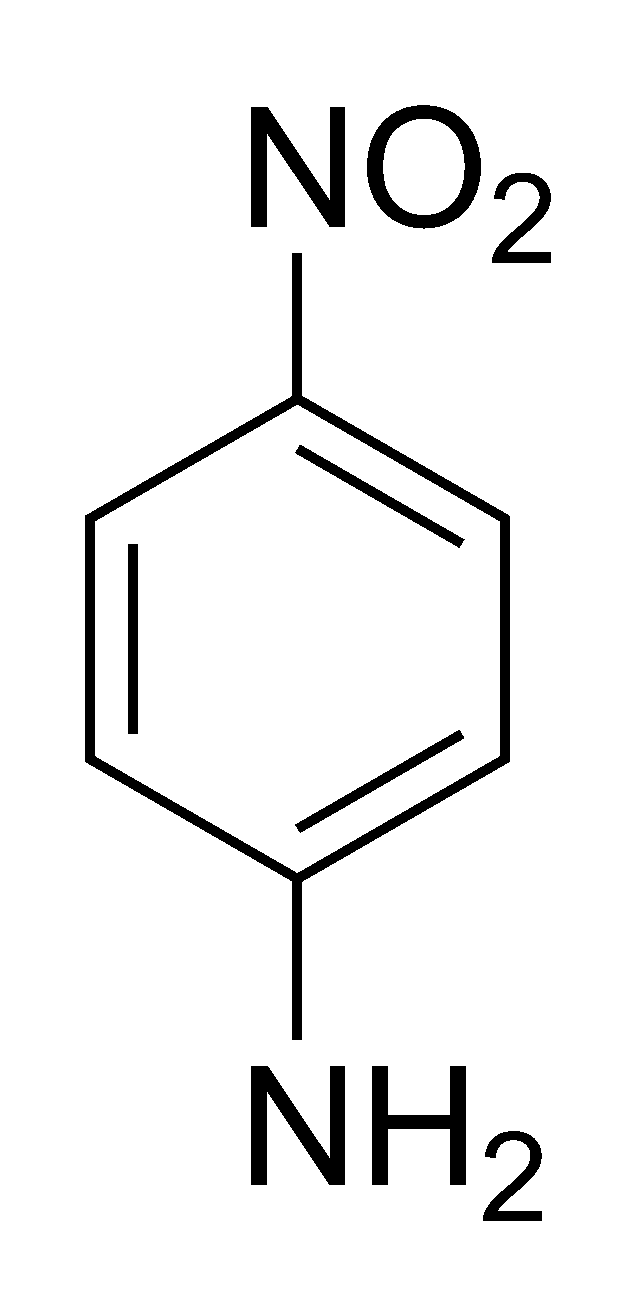
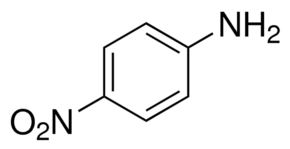
Arrange the following:In increasing order of basic strength:(a) Aniline, p - nitroaniline and p - toluidine

There is a large difference in the melting points of o-nitoaniline and p- nitroaniline. Why? Explain on the basis of different hydrogen bonding (intramolecular v. intermolecular) possible in each compound. | Homework.Study.com