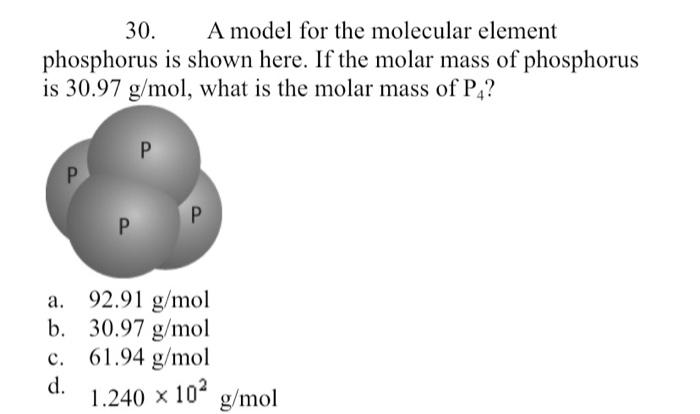
Calculate the molar mass of the following substances. (a) Ethyne, `C_(2)H_(2)` (b) Sulphur molec... - YouTube

Type the correct answer in the box. What is the mass of one mole of phosphorous (P) atoms? Express your - Brainly.com
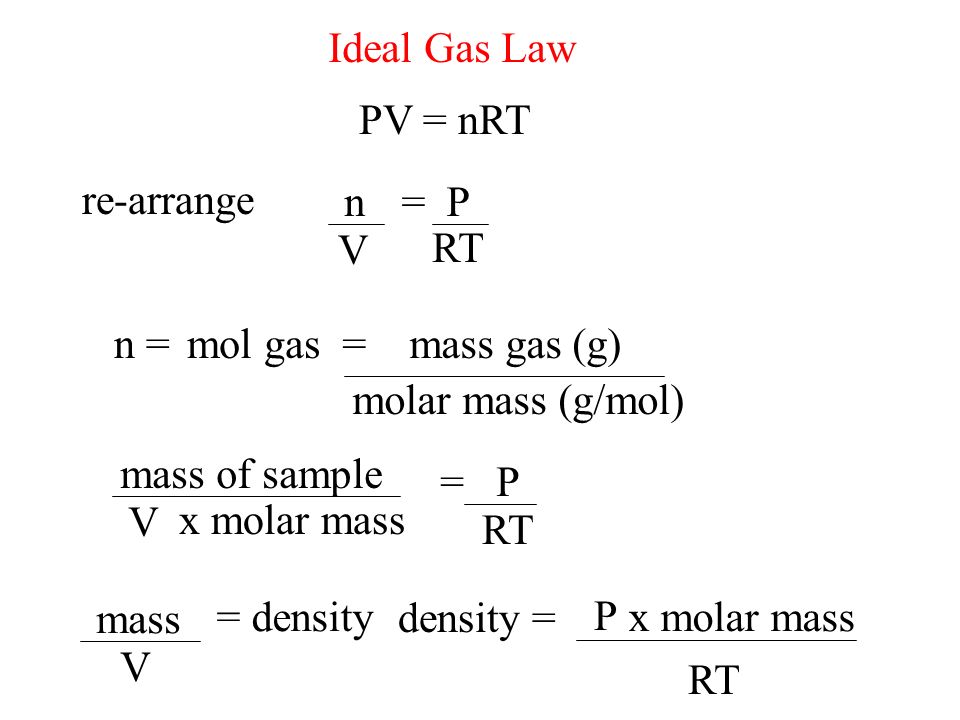
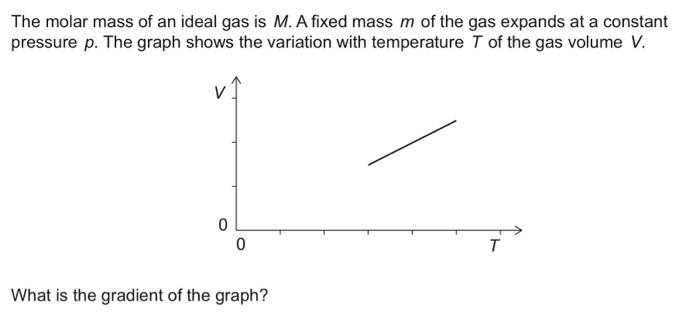
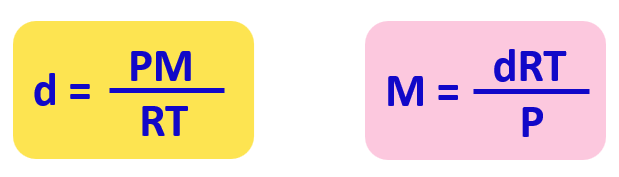
Ideal Gas Law PV = nRT re-arrange n V = P RT n = molar mass (g/mol) mol gas= mass gas (g) mass of sample V x molar mass = P RT =
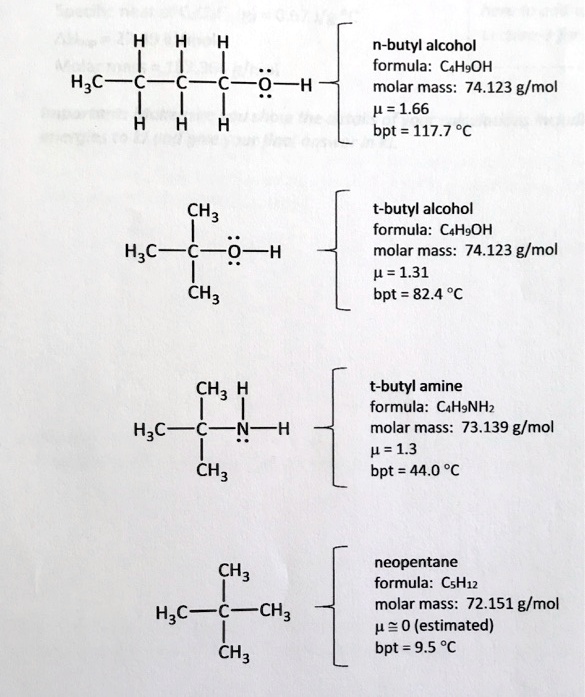
SOLVED: n-butyl alcohol formula: CHsOH molar mass: 74.123 g/mol p =1.66 bpt = 117.7 % H;C H t-butyl alcohol formula: CaHgOH molar mass: 74.123 g/mol p =1.31 bpt = 82.4 % CHz

34.05 mL of phosphorus vapour weighs 0.0625 g at 546 ^∘C and 0.1 bar pressure. What is the molar mass of phosphorus?

SOLVED: Which of the following is the best qualitative graph of P versus molar mass of a [-g sample of different gases at constant volume and temperature? Select one: none 0ithese