
Sips32v8 P Kb-5150 Cem-1 Power Supply Board Professional Equipment Power Support Board Tv Hkc L32a7 Original Power Supply Card - Circuits - AliExpress
![SOLVED: Usefulequations: pH pKa + log ([conjV[WA]) where WA = weak acid pOH = pKb + log ([conjV[WB]) where WB weak base pH = -log1o[HgOt]; [H3Ot] = 10-pH pOH = log10[OH-]; [OH:] = SOLVED: Usefulequations: pH pKa + log ([conjV[WA]) where WA = weak acid pOH = pKb + log ([conjV[WB]) where WB weak base pH = -log1o[HgOt]; [H3Ot] = 10-pH pOH = log10[OH-]; [OH:] =](https://cdn.numerade.com/ask_images/e8201b5be62a4380a6d959f8ef534509.jpg)
SOLVED: Usefulequations: pH pKa + log ([conjV[WA]) where WA = weak acid pOH = pKb + log ([conjV[WB]) where WB weak base pH = -log1o[HgOt]; [H3Ot] = 10-pH pOH = log10[OH-]; [OH:] =

pka & pkb definition and formula |How to find the Strength of an acid and base by pka & pkb values - YouTube
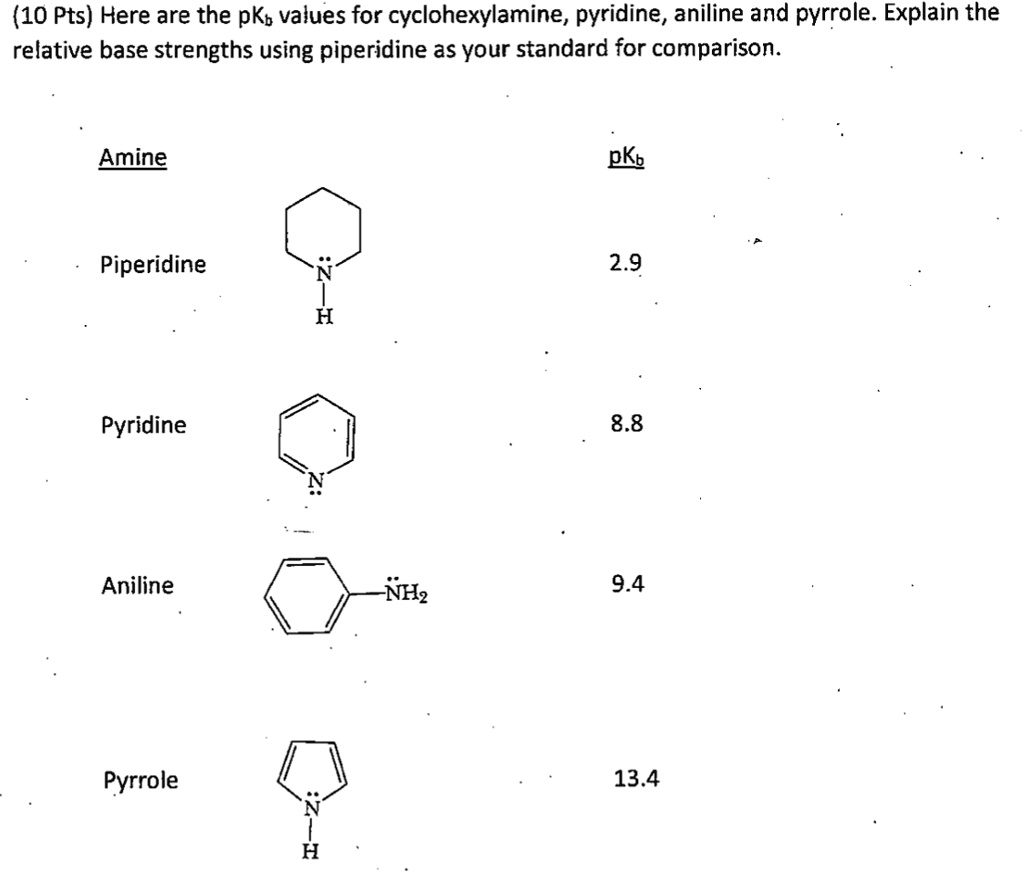
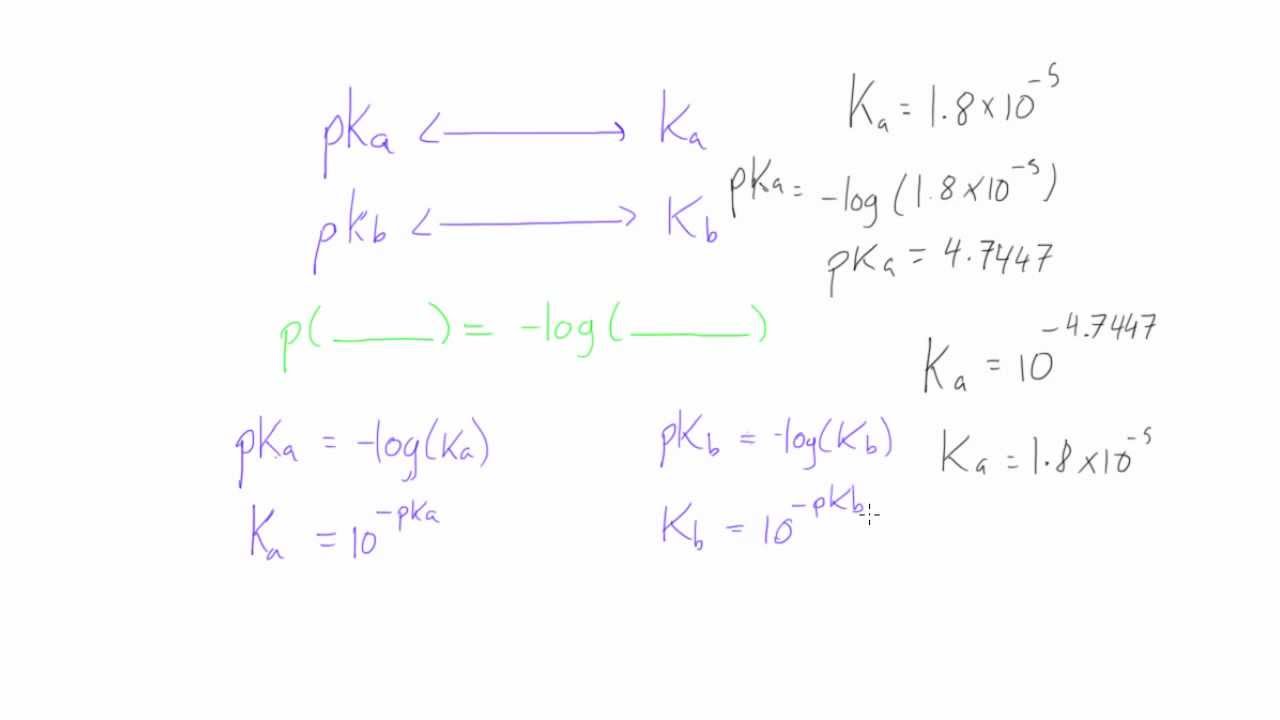













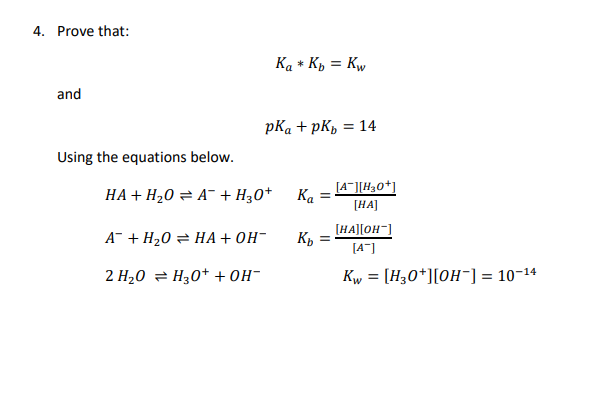
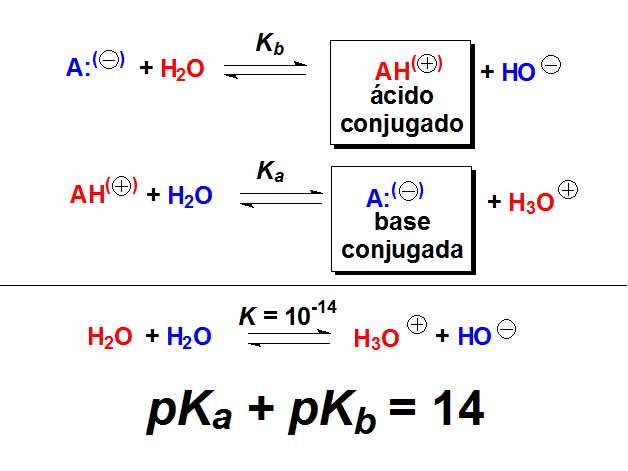

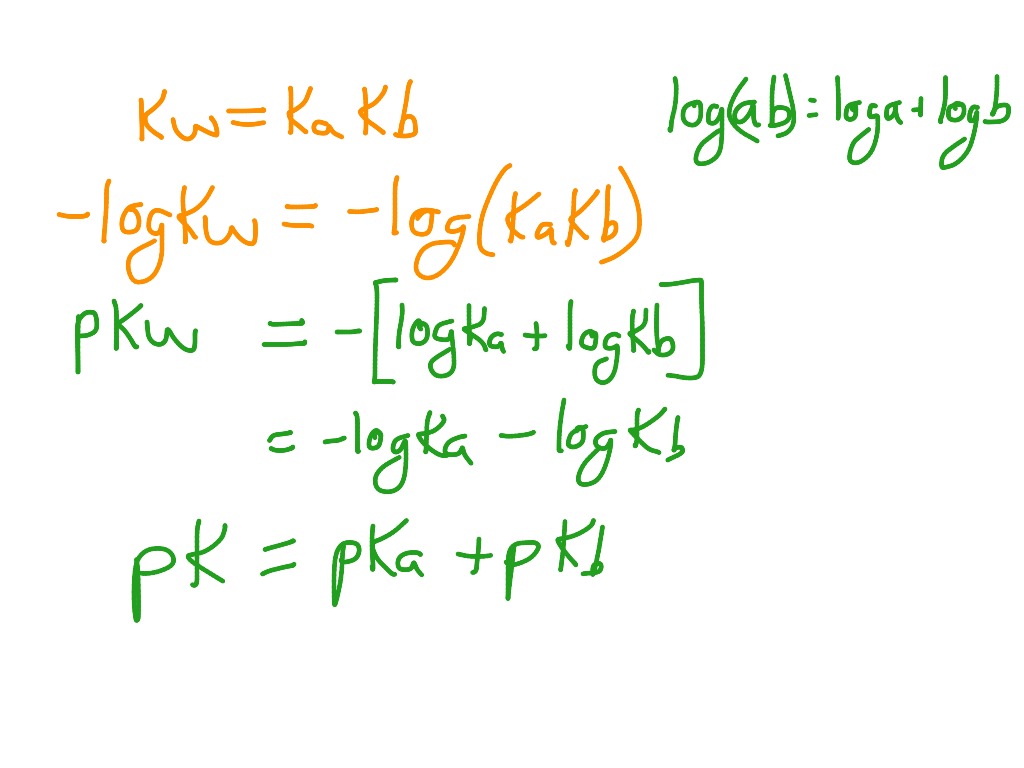
![Solved Important equations pH = pKa + log[salt]/[acid] | Chegg.com Solved Important equations pH = pKa + log[salt]/[acid] | Chegg.com](https://media.cheggcdn.com/media/14b/14b35f43-4468-42d2-a7e2-d924b3fd6eba/phpQPtBXO.png)



